Combating the rise of the superbugs: The health and scientific challenges of antibiotic resistance

It’s hard to imagine the world prior to antibiotics, a world where even a deep laceration could frequently spell significant illness or even death due to infection. Thankfully, since the discovery of penicillin in 1929 by Alexander Fleming, we now have a range of potent antibiotics to treat many of the various types of bacterial infection.
There is a problem though, bacteria are great survivors and have been competing against other bacteria and microorganisms for billions of years. As Professor Matt Cooper from the University of Queensland puts it “Billions of years ago, bacterial species were engaged in an arms race against each other and the chemicals they developed to kill one another have been modified into today’s antibiotics”.1
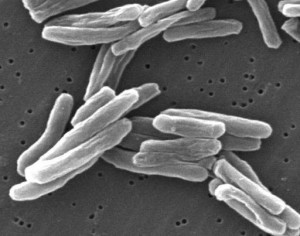
Unfortunately it’s our overuse of these important drugs which has driven the rapid development of antibiotic resistance, the process whereby bacteria containing mutations in their DNA, that provide some protection from an antibiotic, have an enormous survival advantage when exposed to the antibiotic and pretty soon dominate. Frequent exposures to the antibiotic may further strengthen these survival traits via the selection process, rendering the the drug less effective over time. It’s a great example of random variations leading to non-random adaptions through natural selection, although one with profound consequences for human health.
Of particular concern are bacteria that have developed resistance to multiple types of antibiotics, resulting in particularly dangerous resistant bacteria such as the multi-drug resistant variants of tuberculosis, that are extremely difficult to treat. Indeed leading health authorities are so worried about the problem that the Chief Medical Officer of the UK, Professor Dame Sally Davies, has recently labelled the threat as “catastrophic”.2 Addressing the problem of antibiotic resistance is truly one of the biggest challenges of biomedical science and healthcare in the early 21st century.
Why has the pipeline for new antibiotics dried up?
A big area of worry is the lack of new antibiotics in development. This is particularly true for ‘novel’ antibiotics that target new ways combat bacteria.
Part of the problem is that drug development is an increasingly expensive and lengthy process, thus development of medications that have high returns and big impacts, the so called ‘blockbuster drugs’, which focus on the treatment of chronic diseases such as heart disease and diabetes have been highly favored by pharmaceutical companies. After all, people will usually only take an antibiotic course for a week or two, while medications for chronic disease, such as hypertension, are usually needed for a lifetime, meaning much bigger returns for companies. While essential, the focus on development of blockbuster drugs, particularly in 1990s has helped create a void in new antibiotic development.3
Another problem is that development of new agents is a daunting scientific process, thus the focus has been on development of modified versions of existing antibiotics. Novel drugs that look promising in early studies frequently fail for a multitude of reasons, including lack of safety or efficacy in later phases of studies, usually after large investments by pharmaceutical companies, thus giving these companies a case of cold feet for developing novel antibiotics.
Despite this seemingly gloomy outlook, it is not all bad news, for instance drugs that target bacteria in multiple ways rather than using a single mechanism to target them, may provide an important way of helping overcome resistance, at least for a time. Several compounds are in development or being tested using this strategy, for example pharmaceutical company Theravance Inc. has a drug called TD1792 currently in development using this multi-target mechanism.
However, while development of new antibiotics that can target resistant bacteria are essential, it really is only the tip of the iceberg in terms of how to combat antibiotic resistant bacteria
Misuse of antibiotics
A key component to combating antibiotic resistance seems simple enough: reduce their misuse and aim for the rational use of antibiotics. However, as is often the case, doing this is in reality more difficult than at first it might appear.
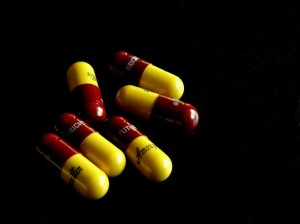
While antibiotics are essential tools in combating many bacterial infections, there are many instances where they are overused. For instance, the overuse of the common antibiotic amoxicillin for the treatment of common acute lower respiratory infections, such as bronchitis, when pneumonia is not suspected has recently been shown to be of limited value in many (but not all) cases and may contribute to development of antibiotic resistance.4
Part of the problem is that Doctors understandably feel pressured to do something for patients and patients may expect a prescription. Particularly important issues here are accurate identification of those who will most benefit from antibiotic use, a difficult task for doctors, with a real need for cheap, rapid and accurate diagnostic tools to classify infections and education of the community to build up greater awareness on the dangers of over prescription.
There’s also the problem of financial benefits for prescribing antibiotics, for instance in China a large number of hospitals rely on pharmaceutical sales for profits, with one study showing that an estimated one quarter of the income made by two of the hospitals investigated was via antibiotic prescriptions.5 In such a profit based environment, risk of inappropriate antibiotic use likely goes up exponentially.
These two examples are really just the tip of the iceberg, however it is obvious that misuse of antibiotics is a problem driven by many factors, including societal, financial and political forces, that will really take improved education and strong regulatory action to overcome.
One thing is for sure: if misuse of antibiotics continues, bacteria will overcome newer multibillion dollar novel antibiotics just as surely as they have overcome other antibiotics.
What can be done?
While development of new antibiotics, and a more rational use of our available antibiotics may be viewed as cornerstones to reducing the spread of antibiotic resistant ‘superbugs’, many other areas can be improved, these include enhanced use of vaccines to reduce need of antibiotic intervention and decreased use of antibiotics in livestock.

Infection prevention should not be overlooked as it is one of the most important strategies to for dealing with antibiotic resistance. Simple hand washing at key times during patient care in hospital has proved highly effective in reducing infection spread, particularly of Bacteria such as Methicillin-Resistant Staphylococcus Aureus (MRSA, sometimes referred to as “golden staph’).6 As an adjunct to highly effective low tech approaches, high technology approaches are also being introduced. John Hopkins hospital in the US recently reported that a trial using high-tech robotic hydrogen peroxide vapour dispensing machines to clean hospital rooms, reduced the number of multi-drug resistant infections by 64%-as a result of these results, the hospital is now two purchasing machines.7
Initiatives which involve collaboration between governments, pharmaceutical companies, researchers and community groups are particularly important in combating resistance, with Europe in particular leading the way in collaborative projects to deal effectively with antibiotic resistance.3
The problem of antibiotic resistance is a global problem that affects all nations, both developed and developing. Thankfully, there is a growing realisation that urgent action has to be taken right now to stem this growing problem. To not act effectively on this issue is to risk going back to dark days when even minor infections would frequently turn deadly.
References:
1. How to stop superbugs in Antibiotic Awareness Week. Available online: http://www.uq.edu.au/news/index.html?article=25544
2. New wave of ‘superbugs’ poses dire threat, says chief medical officer. Available online: http://www.guardian.co.uk/society/2013/mar/11/superbugs-antibiotics-bacterial-diseases-infections
3. Theuretzbacher U 2012. Accelerating resistance, inadequate antibacterial drug pipelines and international responses. International Journal of Antimicrobial Agents 39;295-299.
4. Little P, Stuart B, Moore M, Coenen S, Butler CC, Godycki-Cwirko M 2012. Amoxicillin for acute lower-respiratory-tract infection in primary care when pneumonia is not suspected: a 12-country, a randomised placebo-controlled trial. The Lancet Infectious Diseases 13(2); 123-129.
5. Laxminarayan R, Heymann DL 2012. Challengers of drug resistance in the developing world. British Medical Journal 344:1567.
6. World Health Organisation 2012. The evolving threat of antimicrobial resistance: options for action. Available online: http://www.who.int/patientsafety/implementation/amr/publication/en/index.html
7. Passaretti CL, Otter JA, Reich NG, Myers J, Shepard J, Ross T et al. 2013. An evaluation of environmental decontamination with hydrogen peroxide vapour for reducing the risk of patient acquisition for multidrug-resistant organisms. Clinical Infectious Diseases 56(1); 27-35.
Borradale D (2013-03-14 00:01:17). Combating the rise of the superbugs: The health and scientific challenges of antibiotic resistance. Australian Science. Retrieved: Apr 28, 2026, from https://ozscience.com/biology/combating-the-rise-of-the-superbugs-the-health-and-scientific-challenges-of-antibiotic-resistance/
 Follow
Follow